Which of the Following Has the Highest Ph
A 040 M HCOOH b 040 M HClO 4 c 040 M CH3COOH. View the full answer.

0 1 M Solution Of The Following Has The Highest Ph Youtube
The Ka is called the acid dissociation constant.
. The range of pH and pOH is from 0-14. Cl is a salt of weak base and strong acid. NaNO3 is a salt of strong base and strong acid so pH of the solution will be 7.
View solution View more. Asked Jun 13 2019 in Chemistry by AasaSinha. Which of the following has the highest pH.
View solution The highest p H 1 4 is shown by. Step 1 of 5. 3so the answer is D.
The higher the Ka the higher the hydrogen ion concentration and the lower the pH. 1 M HCl B. Equimolar solution of the following were prepared in water separately.
So sodium bicarbonateis less basic than sodium carbonateis. Among the given solutions NaOH is strongest base. 2 Bile in the gallbladder.
01 M HCl D. The acid is followed by its K a value. Which one of the solutions will record the highest pH.
As concentration of H decreases pH increases and vice-versa. HCN has the highest pH among all the acids listed in the question. PH is inversely proportional to hydrogen ion concentration.
Hence Na 2. The pH of this solution 88. It shows the extent to which an acid is ionized in water.
Which of the following has the highest pH. They do not undergo hydrolysis in aqueous solution. The Fish Tale Across the Wall Tenths and Hundredths Parts and Whole Can you see the.
These factors are dependent on the temperature of the medium. Correct option is B pHlog H 1. Has highest value on pH scale.
Their aqueous solutions are neutral. Thus the pH of the solution 14 - 2 12. Which of the following body fluids has the highest pH.
0001 M HCl Get the answers you need now. The pH shows the hydrogen ion concentration of water. CLASSES AND TRENDING CHAPTER.
Distilled water is almost neutral. AnswerDExplanationThe highest pH is D because001 M HCL. 00001 M HCL.
Which of the following solutions would have the highest pH. Hydrolysis of potassium acetate a salt of a weak acid and a strong alkali gives a weakly alkaline solution since the acetate ion acts as a weak base. 100 7 ratings for this solution.
Therefore NaOH has highest pH. Assume that they are all 010 M in acid at 25 C. So the highest pH is that of dfracM10NaOH.
The pH and pOH solutions are very important factors. NaCl and KCl are salts of strong acid with strong base. Hence the correct option is C.
001 M HCl C. HC 6 H 5 O 13 10 -10 HClO 2 11 10 -2 HCHO 2 18 10 -4 HNO 2 46 10 -4 HOCl 29 10 -8 Expert Answer 100 9 ratings KC6H5O will have the highest p. 201 M HCL.
Acidity is calculated by taking negative log of hydronium ions. Which of the following has highest pH in water. Ammonium chloride NH 4.
N H 4 O H is a weak base and water saturated with chlorine is acidic. View solution Which of the following solutions has the highest p H. 11 M HCL.
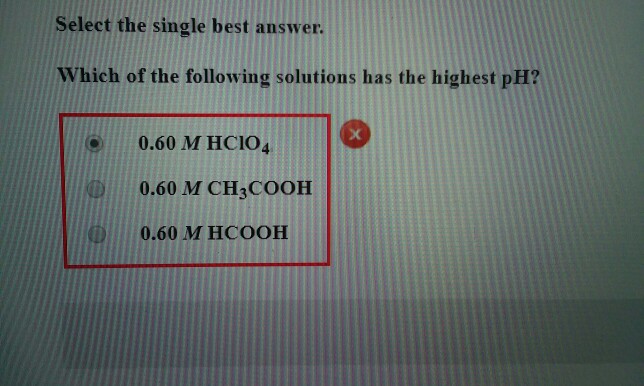
Both a and b have the same pH which is higher than stomach acid. Which of the following solutions has the highest pH.

Solved Select The Single Best Answer Which Of The Following Chegg Com

Which Of The Following Has The Highest Ph Neetlab
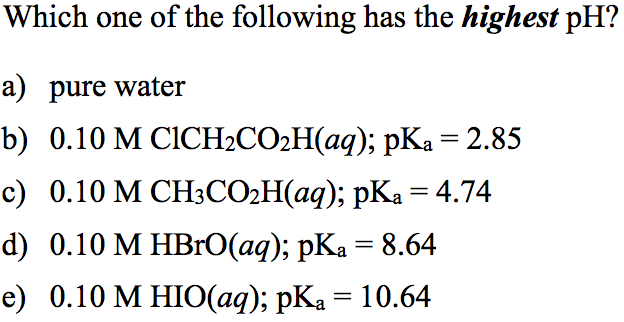
Solved Which One Of The Following Has The Highest Ph Pure Chegg Com
Comments
Post a Comment